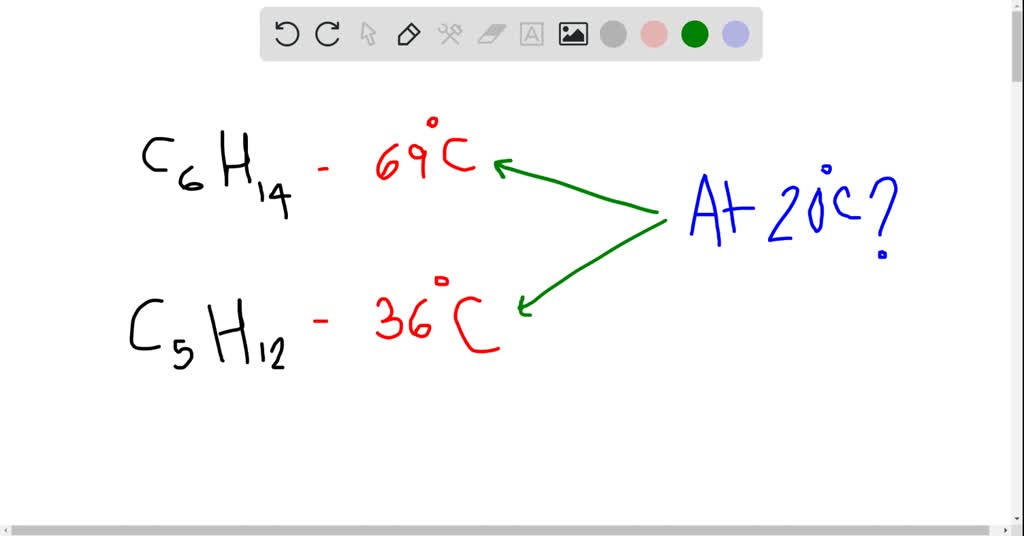
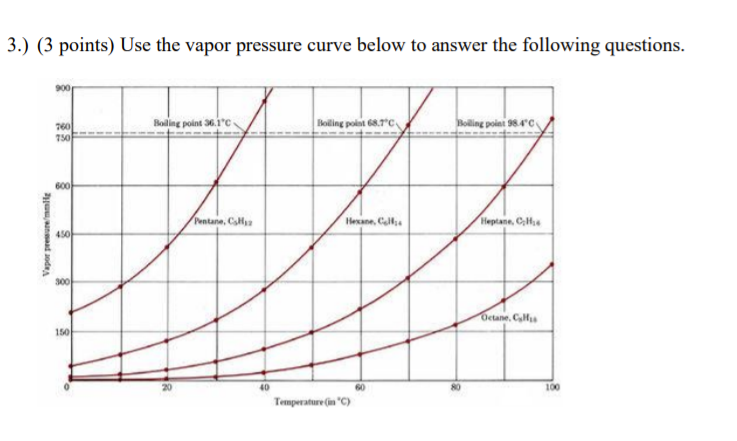
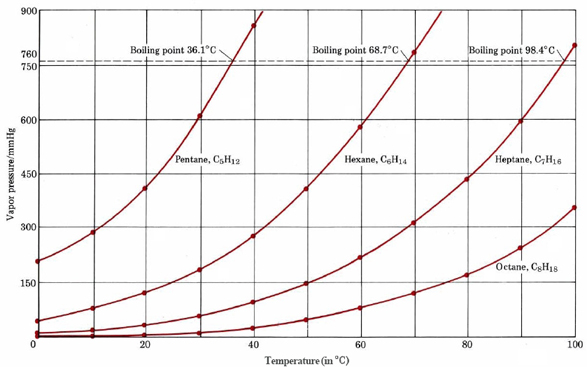
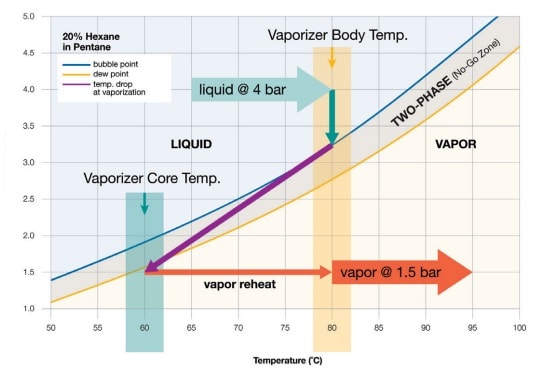
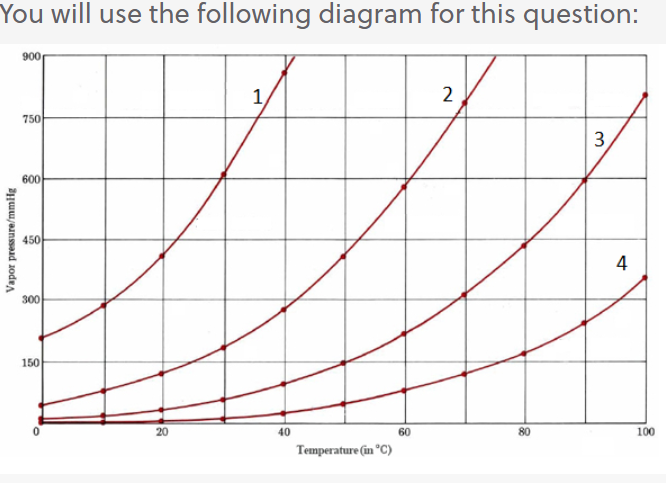
SOLVED: The normal boiling point of hexane is 69 %, and that of pentane is 36 %C. Which of these will have the higher vapor pressure at 20 %C? Hexane Pentane Not

Structural rearrangements in confined n-hexane at elevated temperature. Isobars in pore characterization - ScienceDirect

A solution has a 1:4 mole ratio of pentane to hexane. The vapour pressure of the pure hydrocarbons at 20^∘C are 440 mm of Hg for pentane and 120 mm of Hg

N-hexane vapor pressure curve and the chamber conditions at the start... | Download Scientific Diagram
A solution has a 1 : 4 mole ratio of pentane to hexane. The vapour pressures of the pure hydrocarbons at 20°C - Sarthaks eConnect | Largest Online Education Community

Saturated vapor pressures of CEC gasoline (measured), the 3C mixture... | Download Scientific Diagram