Examining the freezing process of an intermediate bulk containing an industrially relevant protein - ScienceDirect

Breaking old habits: Moving away from commonly used buffers in pharmaceuticals - European Pharmaceutical Review
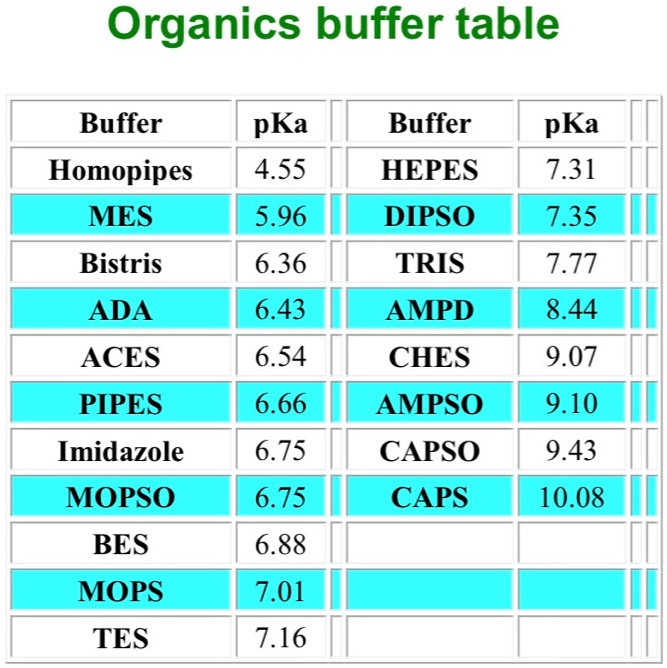
SOLVED: Organics buffer table Buffer Buffer Homopipes MES pKa 4.55 5.96 HEPES pKa 7.31 7.35 DIPSO Bistris 6.36 TRIS AMPD 7.77 ADA 6.43 8.44 ACES PIPES 6.54 CHES AMPSO 9.07 6.66 9.10 9.43 Imidazole 6.75 CAPSO CAPS MOPSO 6.75 10.08 BES 6.88 MOPS TES 7.01 ...

Examining the freezing process of an intermediate bulk containing an industrially relevant protein - ScienceDirect

Table 1 from Measurement of pHT values of Tris buffers in artificial seawater at varying mole ratios of Tris:Tris·HCl | Semantic Scholar

Table 5 from Buffer Standards for the Biochemical pH of the Zwitterionic Buffer N-Tris-(Hydroxymethyl) Methyl-2-Aminoethanesulfonic Acid (TES) from 5℃ to 55℃ | Semantic Scholar

Ciba Foundation Symposium on the Regulation of Cell Metabolism. Cell metabolism. Role of TPN in Control of Glycolysis 235 Table I Phosphatase activities of liver mitochondria Mitochondria Pi liberated xpt. (200

SOLVED: Prepur One of the buffers will be phosphate Or citrate buffer; and the other will be one of the others (not phosphate or citrate) 1. Using the following table, choose the
![Buffer standards for the physiological pH of the zwitterionic buffer 3-[<i>N</i>-tris(hydroxymethyl)methylamino]-2-hydroxypropanesulfonic Acid (TAPSO) From (278.15 to 328.15) K Buffer standards for the physiological pH of the zwitterionic buffer 3-[<i>N</i>-tris(hydroxymethyl)methylamino]-2-hydroxypropanesulfonic Acid (TAPSO) From (278.15 to 328.15) K](https://www.scirp.org/html/7-7100095/2757c7b5-a88d-4273-80de-40545060cb76.jpg)







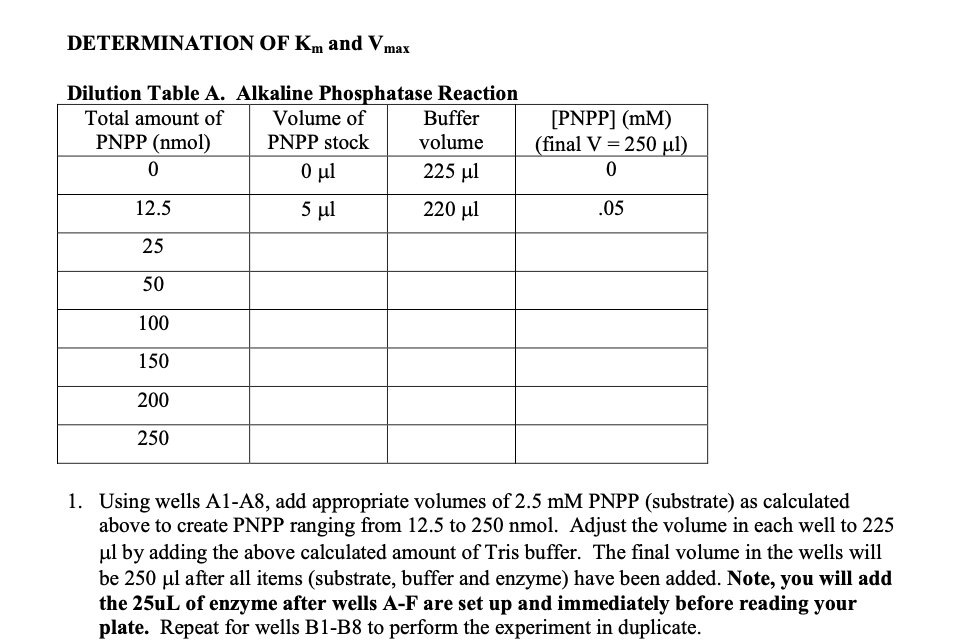


![BT100] 20mM Tris-HCl, pH 8.0 | Biosolution BT100] 20mM Tris-HCl, pH 8.0 | Biosolution](http://biosolution.cafe24.com/wp-content/uploads/2015/07/BT066-Tris-HCl.jpg)