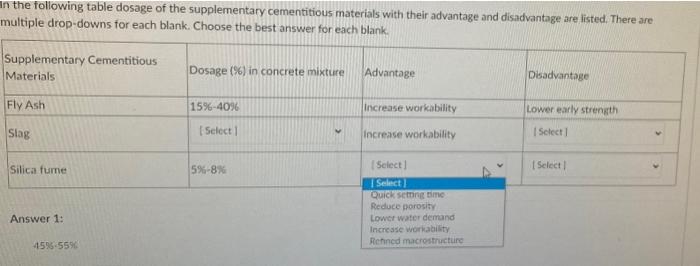
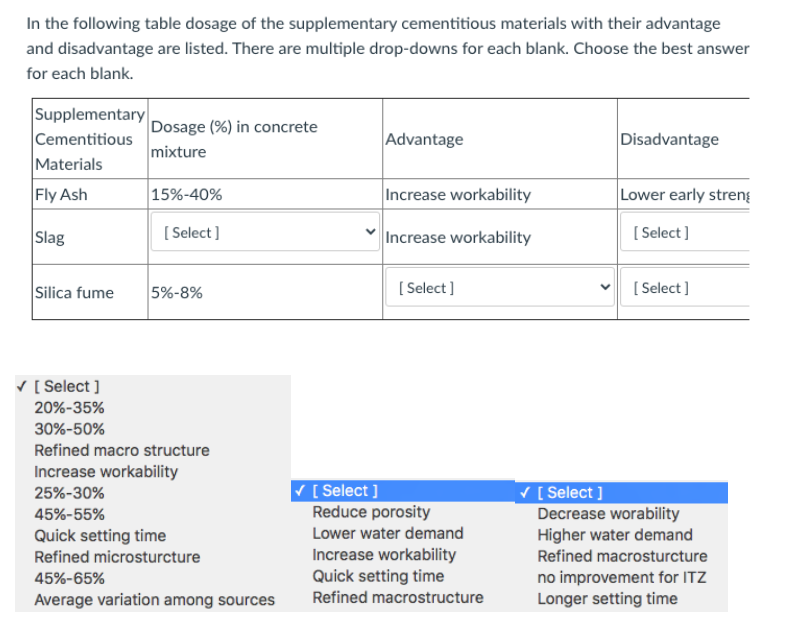
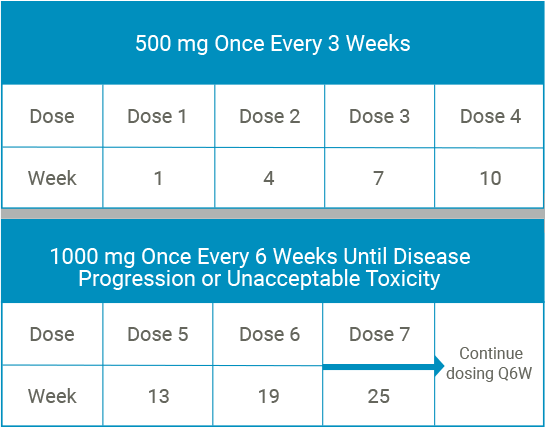
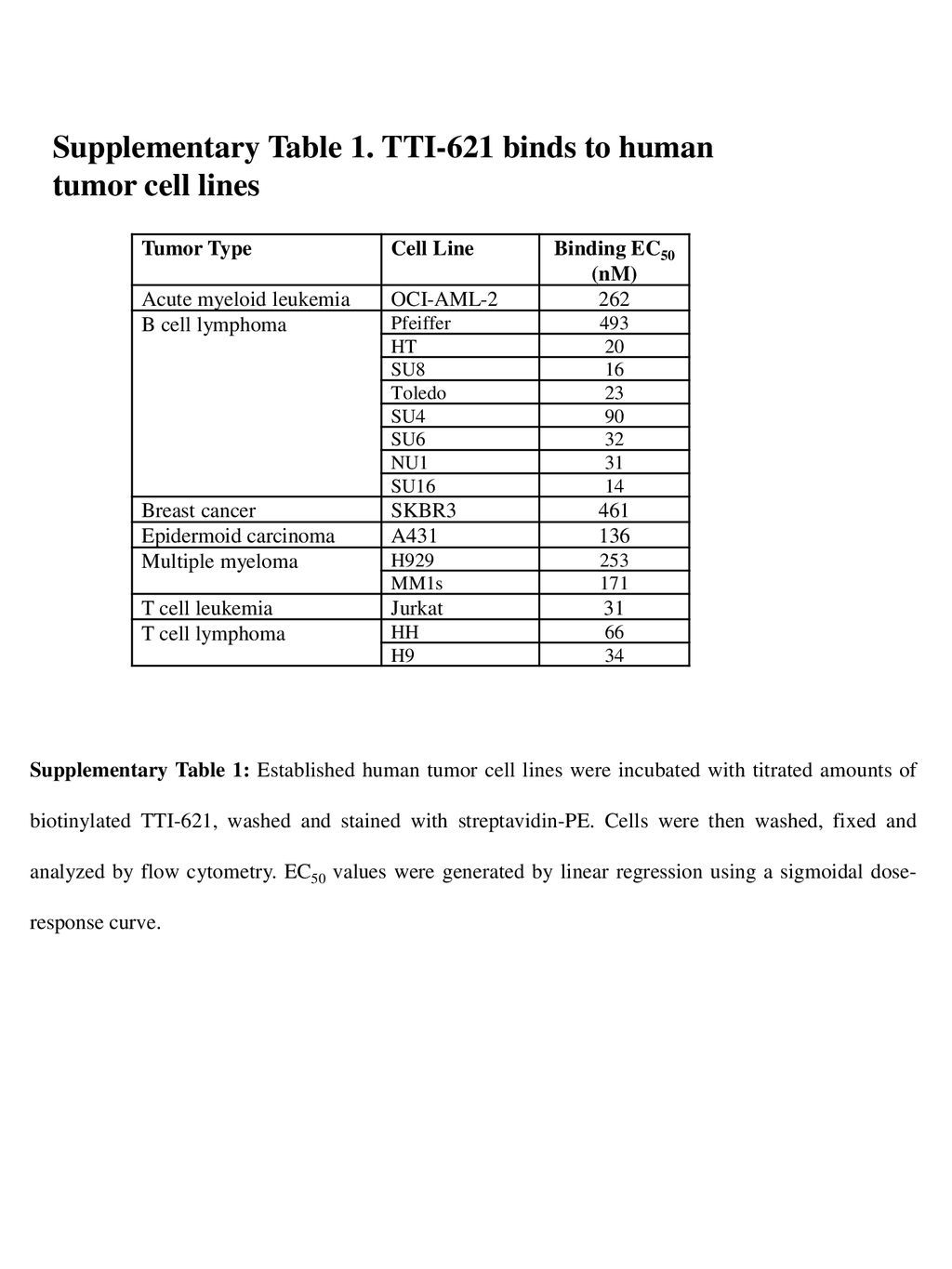
Online Supplementary Table S1 Table S1. Main inclusion/exclusion criteria Main inclusion criteria: Patients eligible for inclus
SUPPLEMENTARY DATA Supplementary Table 1. Model assumptions for the assignment of treatments and estimated therapeutic and behav
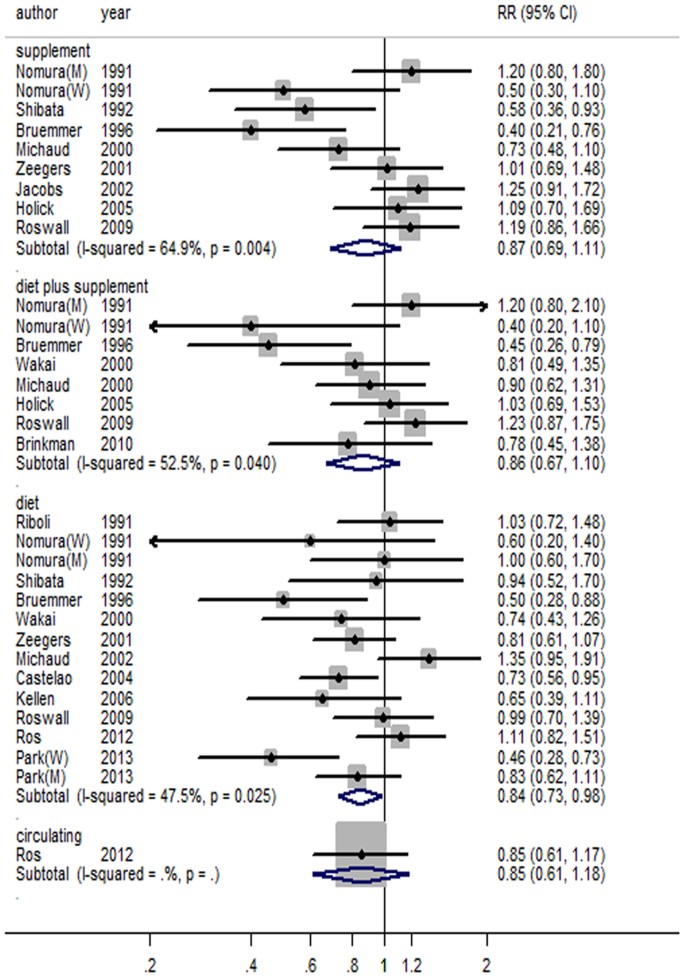
Association of vitamin C, vitamin D, vitamin E and risk of bladder cancer: a dose-response meta-analysis | Scientific Reports
Supplementary Table 2. Criteria for dose delay or dose modification of sorafenib or placebo due to nonhematologic adverse events

Treatment with lacosamide or levetiracetam in patients with renal replacement therapy. What is really known? | Semantic Scholar

Subject flow through the study. See Supplementary Table 1 for details... | Download Scientific Diagram
Supplementary Table 6. Dose changes during the 1-year observa- tion period Discontinuation group Continuation group Total Dose
Supplementary table 1. Information of dabigatran patient for blood collection Dabigatran patient SE Dabigatran dose Twice a day:
Supplementary Table 1 Secondary endpoint changes from baseline after 12 wk of treatment (per protocol set) Variable High dose (n
Supplementary Table S1. Normal tissue constraints for treatment plans Structure Constraints Spinal cord maximum dose <45Gy Ki
List of supplementary Tables and Figures Supplementary Table 1: - Lists of 13 Essential temperature sensitive medicines (8-WHO t