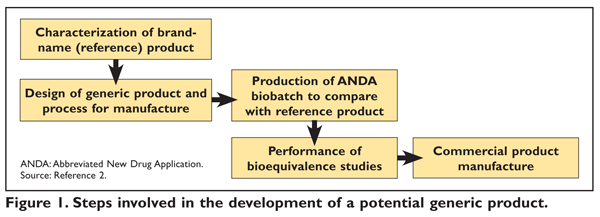
Generic drug development pathways using the current approach to product... | Download Scientific Diagram

Generic drug development pathways using the current approach to product... | Download Scientific Diagram

Pharmaceutical formulation generic Drug in Ghaziabad at best price by Max Care Life Secienc - Justdial
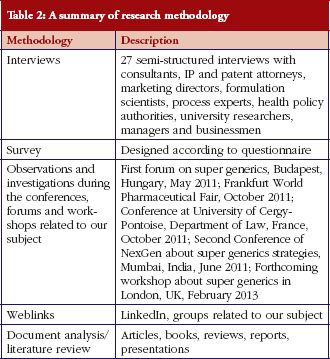
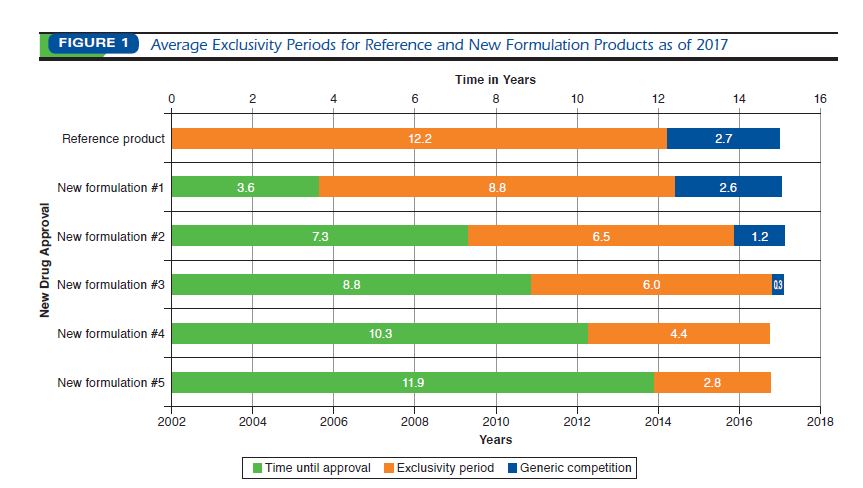
The generic pharmaceutical industry: moving beyond incremental innovation towards re-innovation - GaBI Journal

The Elm | Dec. 6: Center for Research on Complex Generics and FDA Co-Host Free Virtual Public Webinar

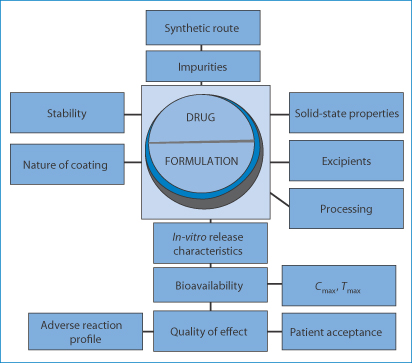
Formulation Development|Sannova Co., Ltd.|Contract Manufacturing organization of Pharmaceutical and Clinical Trial Drugs

Generic Drug Product Development: Solid Oral Dosage Forms, Second Edition (Drugs and the Pharmaceutical Sciences): 9781420086355: Medicine & Health Science Books @ Amazon.com

Comparison of Proprietary and 3 Generic Formulation of Propofol for Induction of General Anesthesia - Pharma Excipients

A pragmatic regulatory approach for complex generics through the U.S. FDA 505(j) or 505(b)(2) approval pathways - Klein - 2021 - Annals of the New York Academy of Sciences - Wiley Online Library

Quality By Design For Generic Drugs Composition Of Formulation Development Experiments | Presentation Graphics | Presentation PowerPoint Example | Slide Templates


![PDF] Safety and efficacy of generic drugs with respect to brand formulation | Semantic Scholar PDF] Safety and efficacy of generic drugs with respect to brand formulation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6f4600551cda3715ef37a0bd9bb9e887e10637a7/5-Table2-1.png)


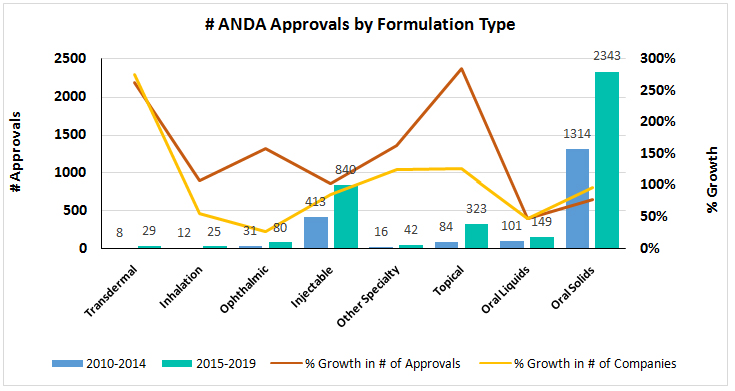

![PDF] Safety and efficacy of generic drugs with respect to brand formulation | Semantic Scholar PDF] Safety and efficacy of generic drugs with respect to brand formulation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6f4600551cda3715ef37a0bd9bb9e887e10637a7/5-Table3-1.png)